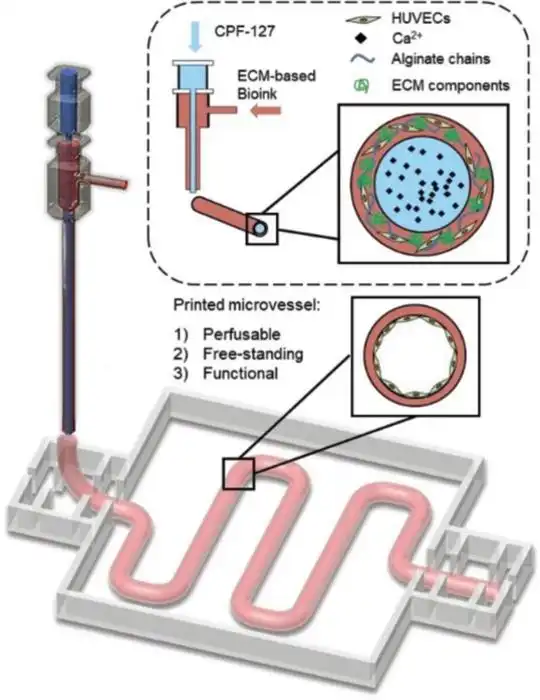
The Bio PRO is Garuda3D's most advanced and fully specified bioprinting system.
Engineered for pharmaceutical R&D departments, clinical research institutes,
and professional tissue engineering centres, it integrates every capability required
for complex, multi-material biofabrication at publication and clinical-grade standards.
The Bio PRO features two independent temperature-controlled Bio-Heads that enable simultaneous
multi-material deposition with precise thermal management per head.
A pneumatic extrusion system replaces conventional stepper-only drives —
delivering smoother, pressure-regulated bioink flow critical for high-viscosity and cell-laden
materials.
The HEPA-filtered sterile enclosure enables direct printing with live cells
and primary tissue, eliminating the need to operate inside a biosafety cabinet.
An integrated touchscreen interface provides full on-device control and
real-time parameter monitoring throughout every print run.
3+ Temp-Controlled Heads
Pneumatic Extrusion
HEPA Sterile Enclosure
Touchscreen Control
UV Curing On-Board
Pressure Monitoring
Heated Bed 80°C
USB + SD Card