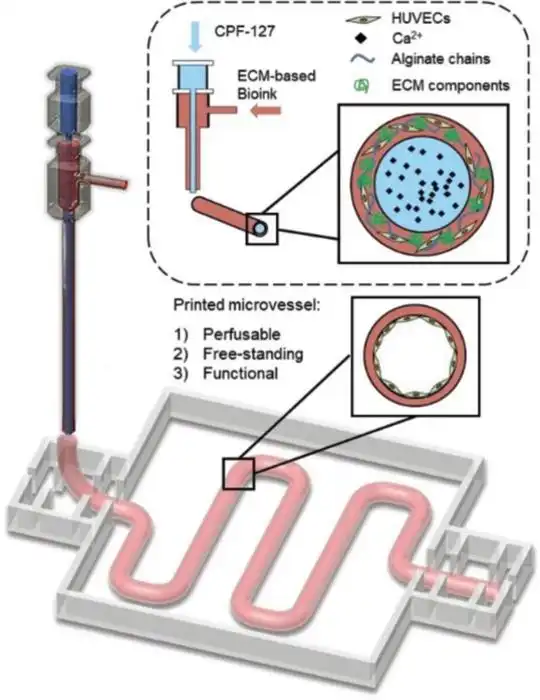
The Bio MORPH is Garuda3D's advanced bio 3D printer, designed for research
organisations that require capabilities beyond standard extrusion-based bioprinting.It retains
the proven Cartesian-frame architecture and BD syringe compatibility of the Bio ATOM
while adding two defining upgrades:
on-board UV curing and real-time pressure monitoring.
These enhancements expand the material palette — photopolymerisable hydrogels such as
GelMA and PEGDA can now be crosslinked in-situ during the print, enabling
complex multi-layer scaffolds with superior structural integrity.
Pressure monitoring ensures consistent volumetric output regardless of bioink batch variation,
making the Bio MORPH the preferred platform for
reproducible, publication-grade bioprinting experiments.
Research Advantage: Pressure data logs can be retained alongside print records,
supporting experimental traceability and enabling correlation between extrusion parameters and construct outcomes.